- Home
- Weddings
- Portraits
- Journal
- Contact
- Pharoah monch simon says instrumental
- Printfab rip screenprinting
- Zan autopurge fallout 3
- Cut the rope character
- Riesling wine
- Hell let loose beta ps5
- Pathfinder wrath of the righteous hilor
- Texas journaly
- Periodic table chemistry referewnce ytables
- Silent sniper rifle hunt badger
- Hermes china

In that same year Englishman Cockroft and the Irishman Walton first split an atom by bombarding lithium in a particle accelerator, changing it to two helium nuclei. This was the complete basis for the periodic table. In 1932 James Chadwick first discovered neutrons, and isotopes were identified. Your chemistry work will most likley use molecular hydrogen (H2) or hydrogen. English physicist Henry Moseley provided atomic numbers, based on the number of electrons in an atom, rather than based on atomic mass. Elements of the periodic table have similarieites across rows and columns. He also transmutated a nitrogen atom into an oxygen atom for the first time. In 1914 Rutherford first identified protons in the atomic nucleus. Radiation is emitted during movement from one orbital to another. In 1913 Bohr discovered that electrons move around a nucleus in discrete energy called orbitals. In 1911 Rutherford and German physicist Hans Geiger discovered that electrons orbit the nucleus of an atom. In 1903 Rutherford announced that radioactivity is caused by the breakdown of atoms. In 1900 Bequerel discovered that electrons and beta particles as identified by the Curies are the same thing. John Townsend and Robert Millikan determined their exact charge and mass. But Mendeleev went one step further than Meyer: He used his table to.

Thomson first discovered electrons small negatively charged particles in an atom. Both published tables with the elements arranged according to increasing atomic mass. In 1894 Sir William Ramsay and Lord Rayleigh discovered the noble gases, which were added to the periodic table as group 0. The Elements In his table, if you were in the same row you had the similar chemical properties thus B.Click the tabs at the top to explore each section General Chemistry: Book Cover They discovered that beta particles were negatively charged. Search: Chemistry Reference Table Workbook. Marie and Pierre Curie started working on the radiation of uranium and thorium, and subsequently discovered radium and polonium.
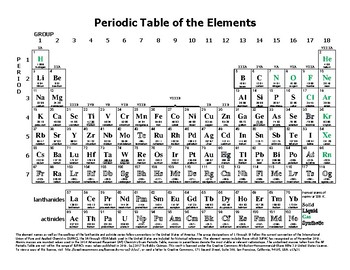
Other Periodic Tables - Links to many periodic tables from the Catalyst. Java Periodic Table - Compare properties with this dynamic table (USC) Graph Chemical Properties - Graph various chemical properties using this resource. Mendeleev, Osnovy khimii (The Principles of Chemistry), Obshchestvenna i a polza: St. Thomson student from New Zealand Ernest Rutherford named three types of radiation alpha, beta and gamma rays. WebElements - Highly interactive periodic table from Mark Winter, University of Sheffield, UK. In 1886 French physicist Antoine Bequerel first discovered radioactivity. He predicted the discovery of other elements, and left spaces open in his periodic table for them. Periodic Videos Tables charting the chemical elements have been around. In 1869 Russian chemist Dimitri Mendeleev started the development of the periodic table, arranging chemical elements by atomic mass. Periodic Table by Royal Society of Chemistry A clickable periodic table with. In 1863 English chemist John Newlands divided the then discovered 56 elements into 11 groups, based on characteristics. In 1809 at least 47 elements were discovered, and scientists began to see patterns in the characteristics. In 1680 Robert Boyle also discovered phosphorus, and it became public. This was the first discovery of phosphorus. He heated residues from boiled urine, and a liquid dropped out and burst into flames. Periodic Table of the Elements Chemistry Reference Sheet California Standards Test Sodium 22.99 Na 11 Atomic number Element symbol Average atomic mass Element name Hydrogen 1.01 H 1 Lithium 6.94 Li 3 Sodium 22.99 Na 11 Potassium 39.10 19 K Nickel 58.69 Ni 28 Rubidium 85.47 Rb 37 Rutherfordium (261) Rf 104 Molybdenum 95.94 Mo 42 Germanium 72. Periodic table history History of the periodic table of chemical elements In 1669 German merchant and amateur alchemist Hennig Brand attempted to created a Philosopher’s Stone an object that supposedly could turn metals into pure gold.
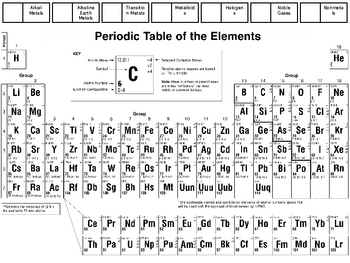
Plant Inspection & Process Optimalisation.Always have the guide available for a quick reference. Navigate from Table of Contents or read page by page - Designed for optimal navigation on electronic devices with a small screen - Use your down time to prepare for an exam. Navigate from TOC or search for words or phrases.FEATURES:- Atomic numbers, symbols & weights - Chemical symbols and more.
PERIODIC TABLE CHEMISTRY REFEREWNCE YTABLES FULL
Periodic Table of the Chemical Elements (Mendeleev's Table) with tables of Melting & boiling points, Density, Electronegativity, Electron affinity, and much more in the Full version.
